SAFETY VISUALIZEDTM
EndoInspect® Lumen Inspection Videoscope System
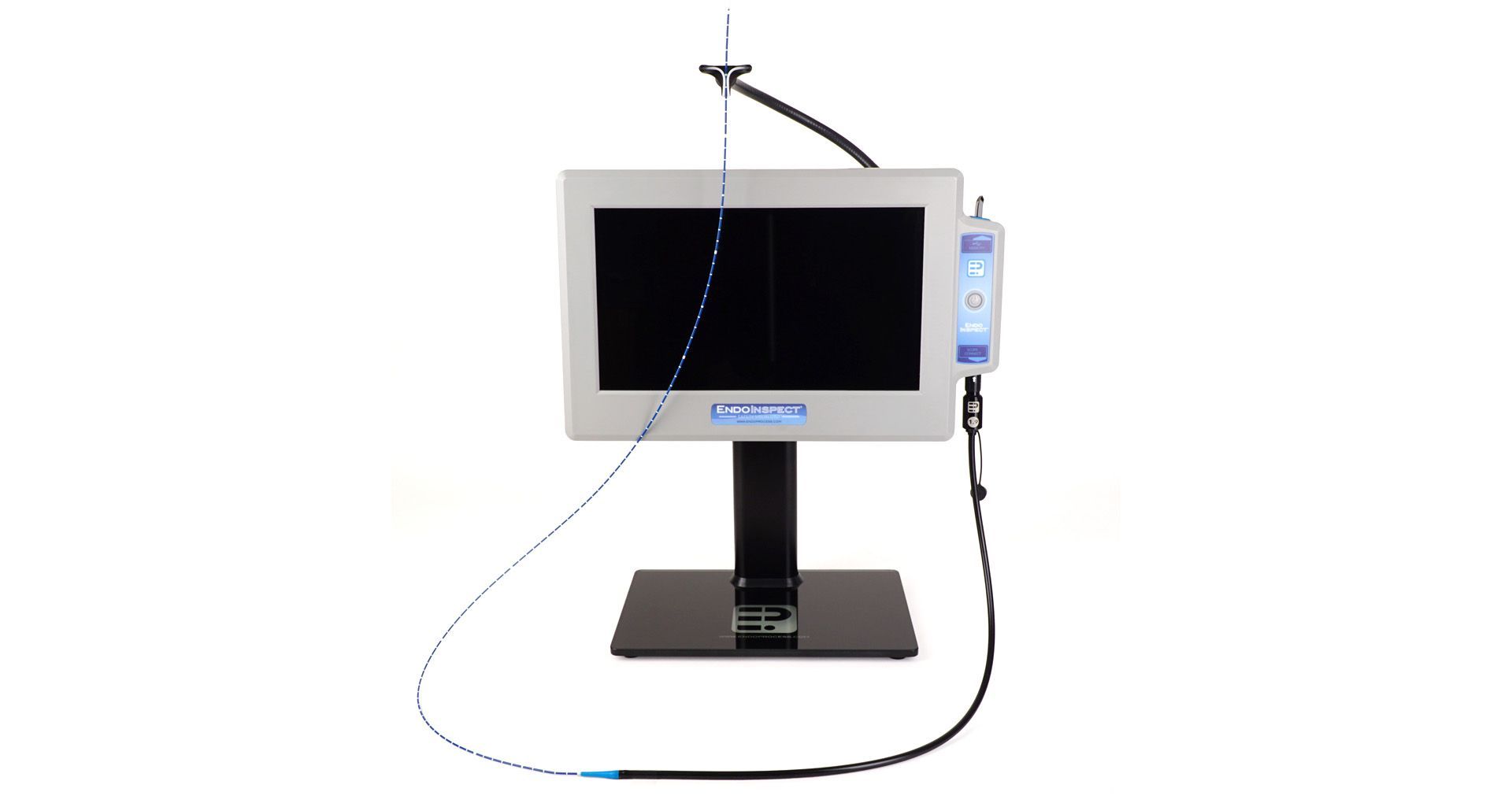
The EndoInspect® lumen inspection system is a modular, plug and play imaging platform technology with a high resolution touch screen video display and interchangeable small diameter inspection scopes. The broad size range of our scope products enables the visual inspection of lumens and devices that have been previously un-inspectable. The EndoInspect® system is IP rated for use in both wet (decontamination) and dry (clean side) environments and all scopes can be fully submerged for high level disinfection (HLD) and cold method sterilization.

Modular Videoscope
Easy to read scope size tag / Rated for HLD liquid submersion
Button
Slide title
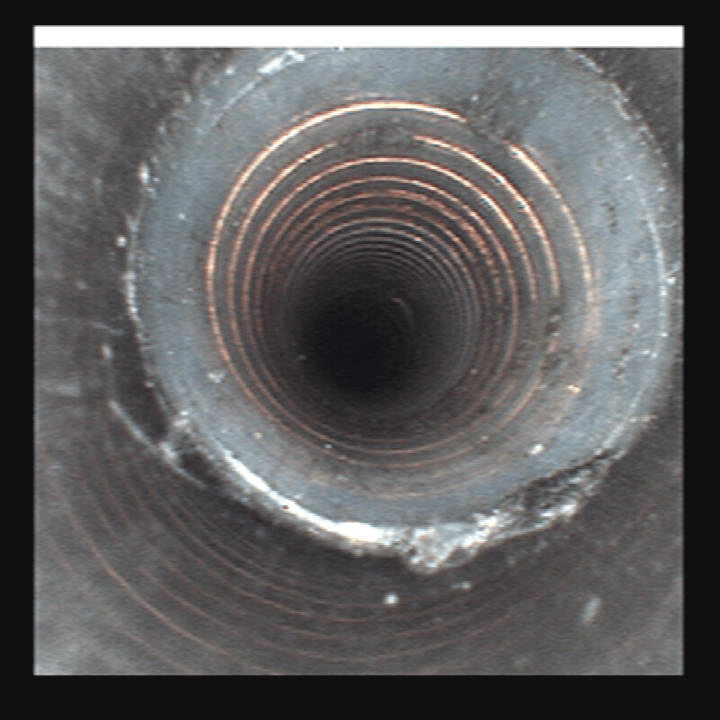
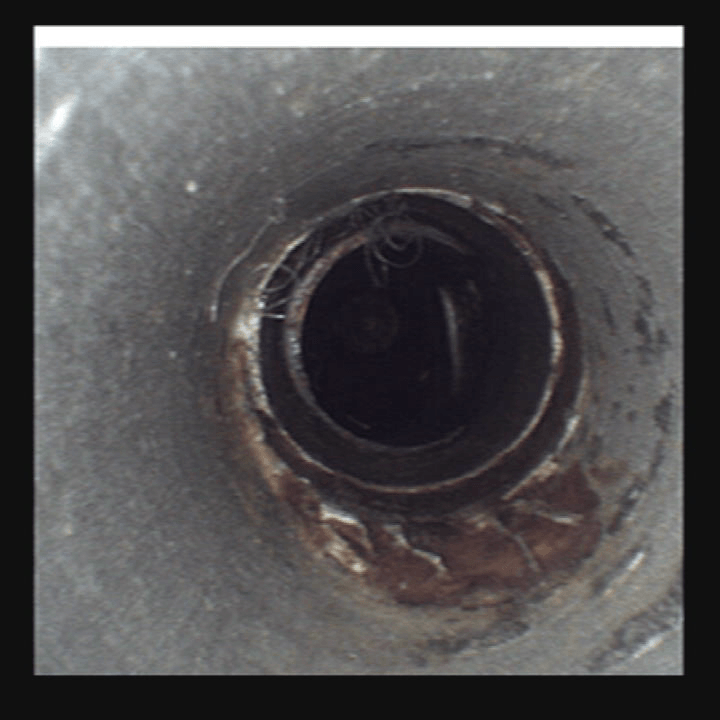
Clear, bright images of the most challanging environments
Button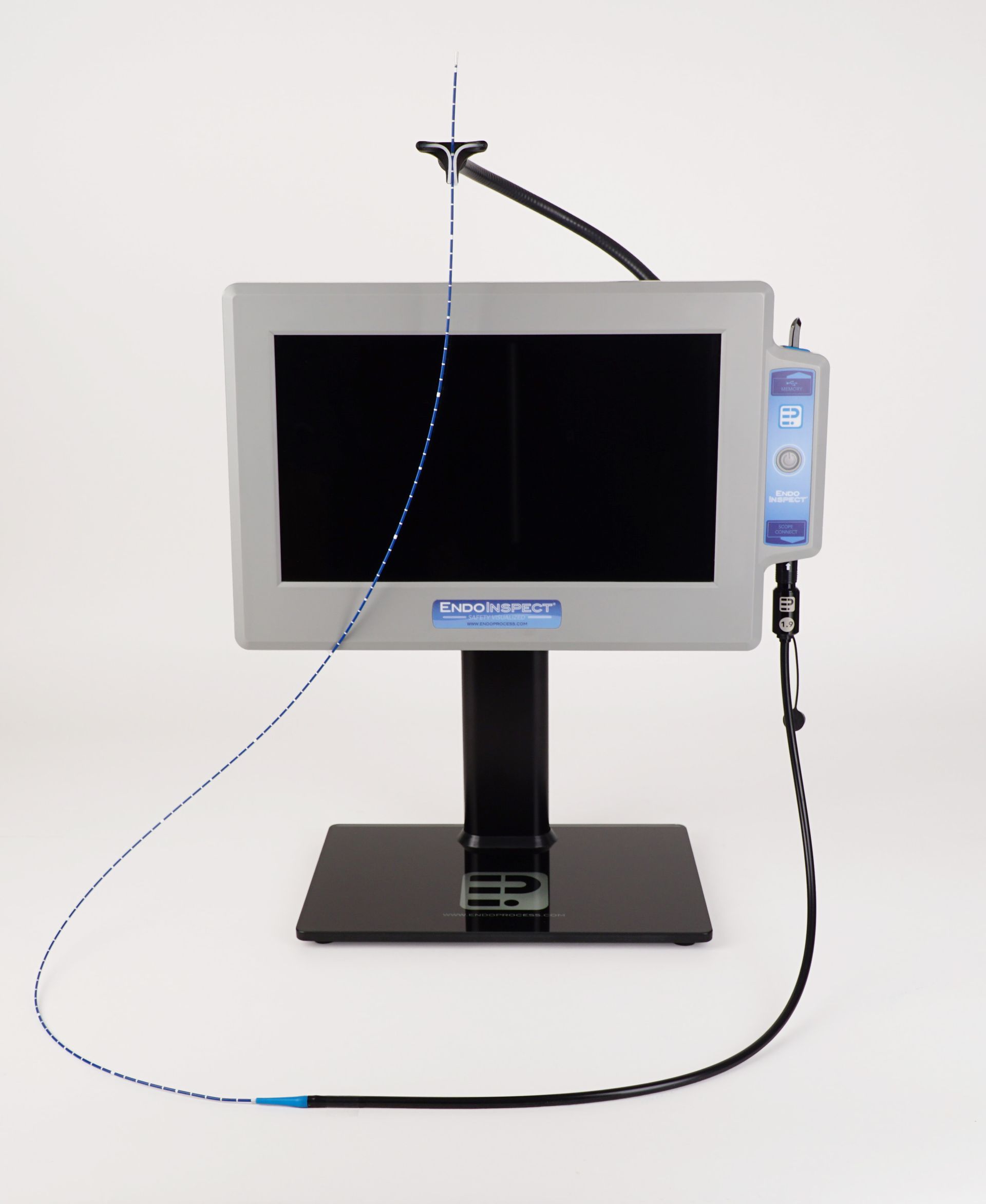
Slide title
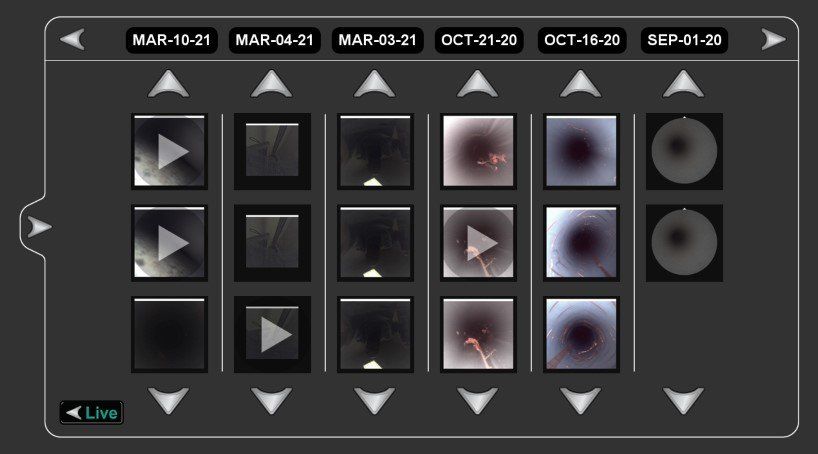
A complete lumen inspection solution with recording capability
Button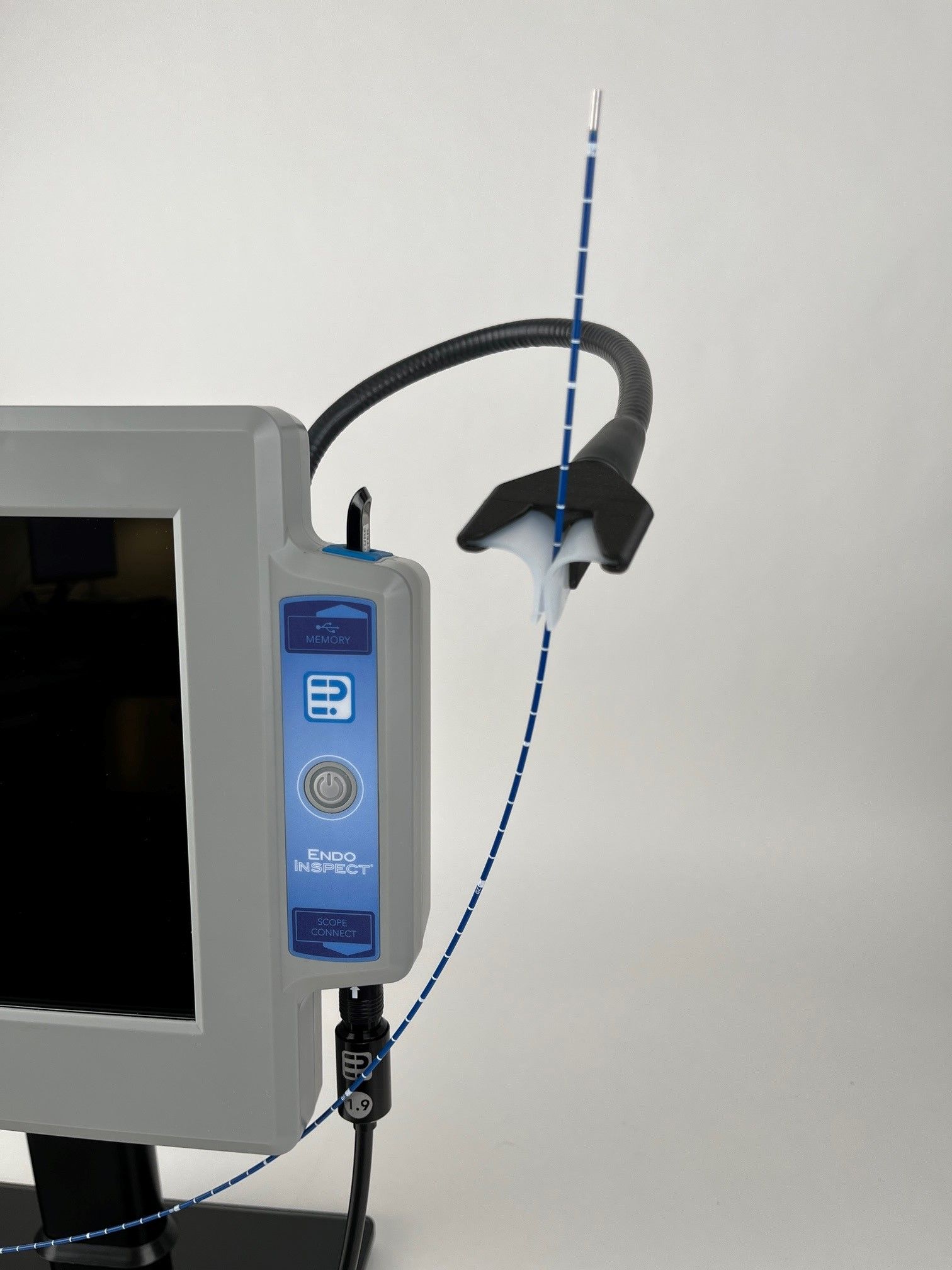
Slide title
Adjustable scope holder
Button
Challenge
Numerous studies have shown that the visual inspection of medical device lumens during decontamination and cleaning prior to final sterilization is a critical step in the success of the Sterile Processing Department workflow. Recommendations calling for the use of visual inspection systems set forth by national organizations such as HSPA, AAMI, AORN, CDC and SNGA (use web links for these) are meant to increase Sterile Processing Department technicians ability to deliver documented, validated, clean and safe endoscopes and surgical devices back to the O.R.
Value
The EndoInspect® system is designed for use by Sterile Processing Department and Central Services Technicians in SPD labs, hospital CS labs and other reprocessing centers. The broad range of IP rated scope sizes combined with the systems high quality imaging and documentation capability enables SPD technicians to perform faster, more successful device inspections.
Product Details
EndoInspect® Monitor:
- 13” Capacitive touch screen video display
- IP65 rating / safe for use in decontamination & wet environments
- No computer or software needed to save images & videos
- All scope models are common to one monitor system
- Simple image & video capture to removable USB storage
- On screen touch keyboard for image & video annotation
- Intuitive file organization w/ image & video file review
- Adjustable, flex-n-stay scope holder system
- Integrated 100 mm VESA mount system
- Table top stand & wall arm mount systems available
EndoInspect® Scopes:
- High resolution CMOS videoscopes in 1.9 & 1.0 mm ⌀
- Industry’s smallest micro-fiberscopes in .8 & .5 mm ⌀
- Scope lengths up to 1900 mm for single pass inspections
- IP68 rating for full submersion during HLD / soak sterilization
- All scope models are common to one monitor system
- Ergonomic, light weight body design w/ scope holder
- Each scope model diameter clearly marked for quick user ID
- Monitor-based push button image & video capture to removable USB storage
- Automatic white balance feature
- Automatic scope LED light adjustment feature
- Automatic, motion detected LED “sleep mode” feature
ABOUT US
CONTACT US
508-206-8627
info@endoprocess.com
EndoProcess | All Rights Reserved